Clinical Trials Integration
Clinical Trials
Are a living ecosystem sponsors, investigators, regulators, IRBs, and, above all, participants moving in sync to turn discovery into dependable evidence. Kernel’s Clinical Trials Integration unifies these parts into one coordinated journey, from first protocol draft to final database lock. We manage the essentials robust informed consent, GCP-aligned quality oversight, transparent communications and submissions with FDA/JFDA and IRBs, and audit-ready documentation while streamlining multi-site collaboration and ensuring participants receive clear information and consistent follow-up.
Contact UsCustom Clinical Study Design & Start-Up
A Quality-by-Design (QbD) approach is applied to translate scientific intent into an operationally feasible protocol and a start-up plan that accelerates first-patient-in. Critical-to-Quality (CtQ) factors are identified at the outset; eligibility, visit schedules, assessments, and data flows are streamlined to reduce burden while preserving statistical power and patient safety. Governance, risk controls, and validation activities are embedded from day one to ensure regulatory acceptance and inspection readiness, including Title 21 of the U.S. Code of Federal Regulations Part 11 (21 CFR Part 11) considerations for computerized systems.
- Protocol architecture: refined endpoints, estimands, visit schedule, and minimal-burden assessments aligned to study objectives under a QbD framework.
- Operational feasibility: site and patient workload modeling; procedure simplification; inclusion of decentralized/remote elements where appropriate.
- Start-up plan: country/regulatory pathways; Institutional Review Board (IRB)/Independent Ethics Committee (IEC) strategy; contract/budget timelines; document-pack readiness.
- Systems readiness: Electronic Data Capture (EDC), electronic Trial Master File (eTMF), Clinical Trial Management System (CTMS), and Interactive Response Technology (IRT) configuration; role-based access; 21 CFR Part 11-compliant controls.
- Risk management: Risk-Based Quality Management (RBQM) plan; issue logs; decision thresholds; validation and User Acceptance Testing (UAT) traceability.

Site Selection
A Quality-by-Design (QbD) approach is applied to translate scientific intent into an operationally feasible protocol and a start-up plan that accelerates first-patient-in. Critical-to-Quality (CtQ) factors are identified at the outset; eligibility, visit schedules, assessments, and data flows are streamlined to reduce burden while preserving statistical power and patient safety. Governance, risk controls, and validation activities are embedded from day one to ensure regulatory acceptance and inspection readiness, including Title 21 of the U.S. Code of Federal Regulations Part 11 (21 CFR Part 11) considerations for computerized systems.
Participant experience
clear consent materials (including eConsent); travel/reimbursement support; reminder cadence.
Vendor orchestration
centralized call-center or digital partners managed to standardized Key Performance Indicators (KPIs) and quality checks.
Live analytics
weekly funnel dashboards (lead → pre-screen → consent → randomization), diversity metrics, and site-level load balancing.
Recruitment blueprint
audience definition; message testing; channel mix; country-specific compliance guardrails.
Pre-screen & triage
Structured scripts; eligibility algorithms; referral pathways integrated with site workflows.
Patient Recruitment
Recruitment is engineered as a measurable funnel from awareness to consent, minimizing patient burden and site workload. Evidence-based outreach combines site databases, referring-physician networks, digital channels, and community organizations, with pre-screening tools that focus effort on eligible candidates. Equity and accessibility considerations are incorporated to broaden reach while preserving protocol integrity and privacy, including use of electronic Informed Consent (eConsent) where appropriate.
Pre-screen & triage
structured scripts; eligibility algorithms; referral pathways integrated with site workflows.
Live analytics
weekly funnel dashboards (lead → pre-screen → consent → randomization), diversity metrics, and site-level load balancing.
Vendor orchestration
centralized call-center or digital partners managed to standardized Key Performance Indicators (KPIs) and quality checks.
Participant experience
clear consent materials (including eConsent); travel/reimbursement support; reminder cadence.
Documentation & Compliance
Management
Documentation is governed through a controlled Trial Master File (TMF) implemented as an electronic TMF (eTMF) and a formal document-control system to ensure version integrity, traceability, and audit readiness. Regulatory, ethics, safety, and operational records are curated against a predefined index; electronic signatures, access controls, and immutable audit trails are maintained. Periodic quality checks, Corrective and Preventive Action (CAPA) pathways, and vendor-oversight files are instituted to keep the trial inspection-ready throughout its lifecycle.
Decentralized Clinical Trials (DCT)
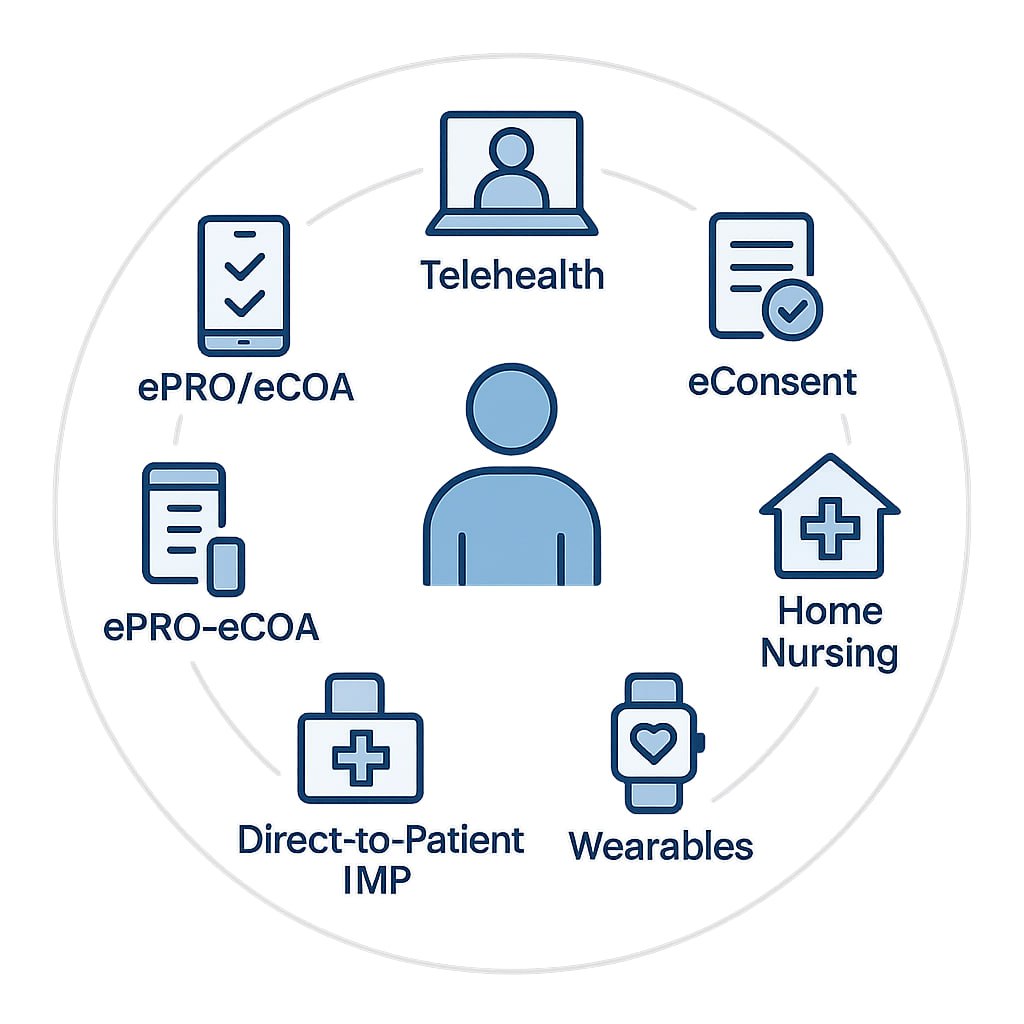
Decentralized clinical trials are studies in which some or all activities happen away from the main investigator site. Typical elements include telehealth, electronic informed consent (eConsent), electronic patient reported outcomes and clinical outcome assessments (ePRO and eCOA), home nursing, local laboratories and pharmacies, wearable sensors, and direct to patient delivery of investigational product. This model lowers participant burden and can expand access while maintaining data integrity and safety under Good Clinical Practice. Clear roles, validated digital tools, secure chain of custody for shipments, and complete documentation are required for inspection readiness.
Our DCT model blends in-person care with remote technologies to speed enrollment, reduce burden, and capture higher-quality, real-world data without compromising GCP, privacy, or safety.
Why Dece ntra lize?
Broader Reach
Higher Retention
What We Deliver ??
Hybrid Study Design
We map each procedure to the optimal setting (home, site, telehealth) and define clear escalation rules for safety.
Participant Services
Home health visits, mobile phlebotomy, drug/device logistics, 24/7 helpdesk, and multilingual support.
Digital Platform Stack
eConsent, ePRO/eCOA, telemedicine, eSource, EDC integration, wearables, and BYOD/Provisioned options—validated and 21 CFR Part 11/Annex 11 compliant.
Data Integrity & Oversight
Risk-based monitoring, audit trails, and real-time quality dashboards aligned with ICH E6(R2)/E6(R3) principles.
Regulatory Navigation
Submission-ready DCT plans, privacy assessments, and localization for MENA and global authorities (e.g., JFDA/SFDA/DHCR/EMA/FDA)
Logistics &Supply Chain
Direct-to-patient (DtP) and direct-from-patient (DfP) workflows with temperature control, chain-of-custody, and IMP accountability.
